Abstract
BACKGROUND Following severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection or vaccination there is significant variability between individuals in protective antibody levels against SARS-CoV-2, and within individuals against different virus variants. However, host demographic or clinical characteristics that predict variability in cross-reactive antibody levels are not well-described. These data could inform clinicians, researchers, and policymakers on the populations most likely to require vaccine booster shots.
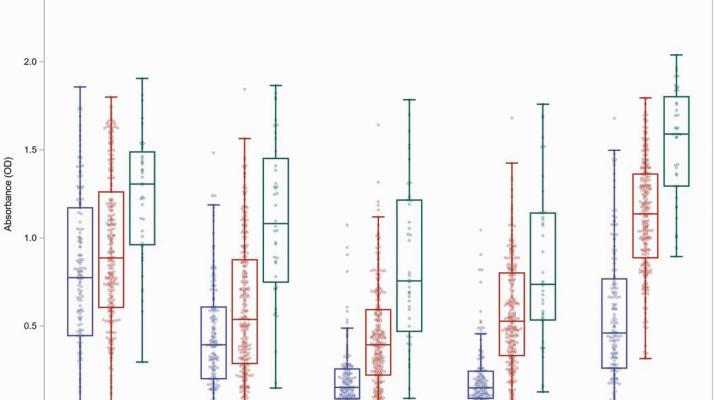
METHODS In an institutional review board–approved prospective observational cohort study of staff at St. Jude Children’s Research Hospital, we identified participants with plasma samples collected after SARS-CoV-2 infection, after mRNA vaccination, and after vaccination following infection, and quantitated immunoglobulin G (IgG) levels by enzyme-linked immunosorbent assay to the spike receptor binding domain (RBD) from 5 important SARS-CoV-2 variants (Wuhan Hu-1, B.1.1.7, B.1.351, P.1, and B.1.617.2). We used regression models to identify factors that contributed to cross-reactive IgG against 1 or multiple viral variants.
RESULTS Following infection, a minority of the cohort generated cross-reactive antibodies, IgG antibodies that bound all tested variants. Those who did had increased disease severity, poor metabolic health, and were of a particular ancestry. Vaccination increased the levels of cross-reactive IgG levels in all populations, including immunocompromised, elderly, and persons with poor metabolic health. Younger people with a healthy weight mounted the highest responses.
CONCLUSIONS Our findings provide important new information on individual antibody responses to infection/vaccination that could inform clinicians on populations that may require follow-on immunization.